“If you know the enemy and know yourself, you need not fear the result of a hundred battles. If you know yourself but not the enemy, for every victory gained you will also suffer a defeat. If you know neither the enemy nor yourself, you will succumb in every battle.” – Sun Tzu from “The Art of War.”
Part 1: Investigating the origin of pandemics
Part 2: Studying genetics and virulence
Part 3: A peek into the viral universe
Part 4: The deepest questions of life
Part 5: Coming to terms with the virus within
Part 1: Investigating the origin of pandemics
In November, doctors in southeastern China began to see the first cases of what would later be a global pandemic. Doctors had never seen the viral illness before, and at first, thought the cases they were seeing might be atypical pneumonia. Even after doctors began to realize that there was something new about the illnesses they were seeing, “it was kept locally for a while, which was one of the problems.” The disease jumped from mainland China to Hong Kong when a medical professor who was infected, checked into a hotel. The professor soon became sick from the illness and went to the hospital, where he died within two weeks. But during his short stay at the hotel, he unwittingly infected several other guests. Those people then took the disease with them to Singapore, Toronto and Hanoi. It soon spread to neighboring areas and then around the world via air travel. Some infected people spread illness more than others do in the same situation, but these superspreaders were a major factor in the spread of this disease. Patients 40 and older who have chronic ailments like those affecting the heart, liver, lungs, and bowel seem to be those who have become sickest. China was criticized and authorities later apologized, for failing to alert world health authorities of the initial outbreak and taking proper precautions to contain it. The mortality rate of this disease was 10%.
The weird and surprising fact is that I was talking about the 2004 SARS pandemic and not the current COVID 19 pandemic. Even the timelines of the COVID and SARS outbreak, starting in November are eerily similar, whether 2004 or 2020. In 2004, a possible source of SARS was identified as a cave full of wild horseshoe bats carrying hundreds of SARS-related viruses. This work, published in a draft paper in 2005, unearthed the link between SARS (severe acute respiratory syndrome) and bats for the first time. The SARS virus seemed to have been adapted to live in bats and not in humans.
The researchers investigating SARS drew heavily upon lessons from two earlier outbreaks.
Hendra virus: In September 1994, a horse trainer in Hendra, Brisbane contracted a virus from his horses. 21 horses were involved and of those, 14 died. Two people caught the disease – a stable hand had a self-curing influenza-like illness, and Vic Rail, a 49-year-old trainer. Both men had very close exposure to the sick horses including nursing and hand feeding them during their illness. The disease, infecting horses and humans, was new to both medicine and veterinary science. Within 7 days of hospitalization, Vic Rail needed ventilation. His condition continued to deteriorate, and he died six days later.
As it happened, one month before the case of Vic Rail, in August 1994, a 36-year-old cane farmer fell ill with an aseptic meningitis-like disease after nursing two of his horses. His horses died, but he recovered. In September 1995 (13 months later) he was readmitted to hospital suffering irritable mood and low back pain with seizures. Over the next week, he developed a fever and more generalized seizures, and despite treatment with anti-bacterials, antivirals, corticosteroids, and anti-convulsants his fever continued, whereupon he lost consciousness and died 25 days after admission.
Somebody noted that the most likely periods for contracting the virus coincides with the birthing season of Australian fruit bat species. By 1996 it had been determined that bats were the most likely natural vector and means of infecting horses. Bats harbored the virus without showing infection, and the virus could be found in a number of tissues in a bat. From the 1990’s onward, it has been suspected that habitat disturbance amplifies the production of Hendra virus by bats. As their habitat disappears, bats are forced to live closer to humans, perhaps the ultimate bad neighbors, and so the opportunity for horses and humans to be exposed to the virus has risen.
Nipah virus: This Malaysian virus was named Nipah after a river in the town where the first victim lived. One afternoon during March 1999 in Singapore, an emergency room doctor had just seen two consecutive patients with fever and confusion and was admitting a third, all of whom were abattoir workers. The same afternoon saw another two workers from the same abattoir with similar symptoms. Later it was learned that a further six abattoir workers had presented to other hospitals the same week, one of whom died after a rapid neurological deterioration. A team of microbiologists at the University of Malaya in the Malaysian capital Kuala Lumpur isolated a paramyxovirus 5 days after obtaining the first patient samples. Researchers studied a large bat colony on Tioman island, off the eastern coast of West Malaysia. After collecting over 1,000 bat urine samples, the virus was detected in the urine of a species of flying fox. The virus was also found in a piece of fruit that had been partially eaten by a bat. This implied the presence of the virus in the animal’s saliva. Because many Malaysian pig farms have fruit trees, it is easy enough to imagine how the virus can be transmitted from bats. Pigs probably serve as amplifying hosts after ingesting infected bat urine, saliva, or discarded food items.
The COVID 19 scare: Reporter Jane Qiu has a nice writeup on the Scientific American:
“In November 2019, mysterious patient samples arrived at Wuhan Institute of Virology at 7 P.M. on December 30, 2019. Moments later, Shi Zhengli’s cell phone rang. It was her boss, the institute’s director. The Wuhan Center for Disease Control and Prevention had detected a novel coronavirus in two hospital patients with atypical pneumonia, and it wanted Shi’s renowned laboratory to investigate. If the finding was confirmed, the new pathogen could pose a serious public health threat—because it belonged to the same family of bat-borne viruses like the one that caused severe acute respiratory syndrome (SARS), a disease that plagued 8,100 people and killed nearly 800 of them between 2002 and 2003. “Drop whatever you are doing and deal with it now,” she recalls the director saying. If coronaviruses were the culprit, she remembers thinking, “could they have come from our lab?” She walked out of the conference she was attending in Shanghai and hopped on the next train back to Wuhan.
To Wuhan based virologist, Shi Zhengli, life came full circle. Her first virus-discovery expedition was on a breezy, sunny spring day in 2004, when she joined an international team of researchers to collect samples from bat colonies in caves near Nanning, the capital of Guangxi. These expeditions were part of the effort to catch the culprit in the SARS outbreak. Shi’s team used the antibody test to narrow down locations and bat species to pursue in the quest for these genomic clues. After roaming mountainous terrain in the majority of China’s dozens of provinces, the researchers turned their attention to one spot: Shitou Cave on the outskirts of Kunming, the capital of Yunnan—where they conducted intense sampling during different seasons throughout five consecutive years.
The efforts paid off. The pathogen hunters discovered hundreds of bat-borne coronaviruses with incredible genetic diversity. “The majority of them are harmless,” Shi says. But dozens belong to the same group as SARS. In Shitou Cave—where painstaking scrutiny has yielded a natural genetic library of bat viruses—the team discovered a coronavirus strain in 2013 that came from horseshoe bats and had a genomic sequence that was 97 percent identical to the one found in civets in Guangdong. The finding concluded a decade-long search for the natural reservoir of the SARS coronavirus.
And by January 7, 2020 the Wuhan team determined that the new virus had indeed caused the disease those patients suffered—a conclusion based on results from polymerase chain reaction analysis, full genome sequencing, antibody tests of blood samples and the virus’s ability to infect human lung cells in a petri dish. The genomic sequence of the virus—now officially called SARS-CoV-2 because it is related to the SARS pathogen—was 96 percent identical to that of a coronavirus the researchers had identified in horseshoe bats in Yunnan, they reported in a paper published last month in Nature. “It’s crystal clear that bats, once again, are the natural reservoir,” says Daszak, who was not involved in the study.
Source: shorturl.at/rKPS8
COVID 19 could be another virus programmed to live in bats and not in humans. It was initially called the SARS COV 2 virus (SARS family of viruses) before being called as COVID 19. There are reports that patient zero in the COVID pandemic was a shrimp seller from the Huanan seafood market who contracted it on November 17, 2019. While paradoxically this place is just a couple of miles away from the Wuhan center of disease control and the Wuhan Institute of Virology, scientists have declared that COVID 19 has natural origins. In addition to bats, there is also research that indicates the source could have been wild pangolins, with a match of 88-92% (the most trafficked wild animal in the world). It is very important to stamp out wildlife consumption by humans to prevent similar outbreaks in the future. On February 24, 2020, China took bold steps to shut down the wildlife market industry worth $76 billion, even though it put approximately 14 million people out of a job. These are unconfirmed reports that the wet markets are up and running again. As of 6 April, this has infected 1.3 million people and caused 72638 deaths, with a mortality rate of 5.5%.
A natural question to ask is why bats are implicated in COVID, SARS and other outbreaks in the past. It turns out that Bats are the natural reservoir all types of dangerous viruses, namely the Ebola, Marburg, Nipah and Hendra viruses. Except for the rabies virus, bats can host all these viruses without becoming sick. So, the question is why do viruses co-exist and tolerate all these dangerous viruses. In a paper published in the journal Cell Host & Microbe in February 2018, scientists at the Wuhan Institute of Virology in China found that the energy demands of bat flight are so great that cells in the body break down and release bits of DNA that are then floating around the body. In most mammals, this would cause serious inflammation as the body would treat DNA particles like viruses. Bats have lost some genes involved in that response, which makes sense because the inflammation itself can be very damaging to the body. They have a weakened response but it is still there. Thus, the virologists write, this weakened response may allow them to maintain a “balanced state of ‘effective response’ but not ‘over response’ against viruses.” The DNA damage repair genes in bats also work more efficiently.
Bats could also harbor a large number of viruses (more than 60 which are harmful to humans alone) because they live in close proximity to each other, and viruses spread quickly in these groups. Since humans also tend to live in groups, viruses primed to live in bats can easily spread to humans. They also have adopted peculiar human traits allowing viruses which live in bats to spread among humans. For example, bats exhibit altruistic behavior, which can serve as transmission routes for viruses. When a bat that has not fed in the night begs for food, other bats, especially females, have no qualms about regurgitating the blood and allowing that unfed bat to feed. Even nursing bats are known to share their milk with males that are hungry. In addition, when a baby bat is orphaned, it is normally adopted by an older female in the colony.
Other researchers have suggested that bats’ super-tolerance might have something to do with their ability to generate large repertoires of antibodies or the fact that when bats fly, their internal temperatures are increased to around 40 deg C, which is not ideal for many viruses. Only the viruses that have evolved tolerance mechanisms survive in bats. These hardy viruses can therefore more tolerate human fever and thrive in humans much better than they would inside bats.
Part 2 – Studying genetics and virulence
COVID 19: Coronaviruses have historically caused mild common colds, outside incidents like the SARS and MERS outbreaks. Mutations in 2019 have turned this less harmful Coronavirus into the more malevolent COVID 19 version. The COVID 19 spike proteins had evolved to effectively target a molecular feature on the outside of human cells called ACE2 , a receptor involved in regulating blood pressure, 10 times more efficiently. This coupled with another mutation to use human enzymes like Furin, to spit its RNA into the cell, makes this virus particularly potent.
The COVID 19 virus seems to have been evolved rapidly in the last few decades. Looking at the diagram below, which depicts mutations and the emergence of new strains over time, the COVID 19 virus is more similar to viruses from Bats than from Pangolins. A mutated version that infected bats or pangolins probably began the human outbreak.

Another interesting fact uncovered by Chinese researchers is that the COVID 19 virus is evolving as we speak, in front of our eyes. After the Wuhan outbreak, the virus has further mutated into 2 types, the L type (70% of cases) and S type (30% of cases). Whereas the L type was more prevalent in the early stages of the outbreak in Wuhan, the frequency of the L type decreased after January 2020. Human intervention may have placed more severe selective pressure on the L type, which might be more aggressive and spread more quickly. On the other hand, the S type, which is less aggressive, might have increased in relative frequency due to relatively weaker selective pressure. Later I argue that viruses that are extremely virulent do not fare as well as viruses that are less virulent. Less virulent viruses can spread and survive in populations longer.
SARS: Looking at the diagram below, the SARS virus infecting humans (SARS 4 and SARS 3) seems closer to viruses infecting civets (SARS 2) more than even those infecting bats (SARS 1 and SARS 5-8). The current thinking is that deadly strain probably originated in China’s horseshoe bats and later passed through masked palm civets sold in Guangdong’s animal markets.

Virulence of viruses: Typically, malignant viruses have less interest in harming their host humans in the long term. Natural selection balances this trade-off, selecting for pathogens virulent enough to produce many offspring (that are likely to be able to infect a new host if the opportunity arises) but not so virulent that they prevent the current host from presenting them with opportunities for transmission. Where this balance is struck depends, in part, on the virus’s mode of transmission.
Sexually-transmitted pathogens, for example, will be selected against if they immobilize their host too soon, before the host has the opportunity to find a new sexual partner and unwittingly pass on the pathogen. Some biologists hypothesize that this trade-off helps explain why sexually-transmitted infections tend to be of the lingering sort. Even if such infections eventually kill the host, they do so only after many years, during which the pathogen might be able to infect a new host. On the other hand, diseases like cholera (which causes extreme diarrhea) are, in many situations, free to evolve to a high level of virulence. Cholera victims are soon immobilized by the disease, but they are tended by others who carry away their waste, clean their soiled clothes, and, in the process, transmit the bacterium to a water supply where it can be ingested by new hosts. In this way, even virulent cholera strains that strike down a host immediately can easily be transmitted to a new host. Accordingly, cholera has evolved a high level of virulence and may kill its host just a few hours after symptoms begin. it suggests how we might sway pathogen evolution towards less virulent strains. In situations where high virulence is tied to high transmission rates (e.g., cholera), reducing transmission rates (e.g., by providing better water sanitation) may favor less virulent forms. The idea is to create a situation in which hyper-virulent strains that soon kill or immobilize their hosts never get a chance to infect new hosts and are turned into evolutionary dead ends. In fact, biologists have observed this phenomenon in South America: when cholera invaded countries with poor water sanitation, the strains evolved to be more virulent, while lineages that invaded areas with better sanitation evolved to be less harmful.
Virulence, and link with evolutionary history: Tracing the history of viruses, tracking their virulence and mutations also gives insight into human evolution, primate evolution, vertebrate evolution and ultimately the evolution of surface life. This becomes evident when studying some viruses like HPV, Herpes, and Hepatitis.
HPV: Human papillomavirus (HPV) is an STD causing virus with just 8 genes. It impacts 79 million Americans, but around 90% of infections cause no symptoms and resolve spontaneously within two years. But the trouble is for the remaining 10%. When a high-risk HPV infection persists for many years, it can lead to cell changes that, if untreated, may get worse over time and become cancer. Around 5.2% of all cancers are caused by HPV. HPV causes about 610,000 cancers annually worldwide, including virtually all cervical cancers, and many anogenital and head and neck cancers.
The major evolutionary separation between some papillomaviruses found only in humans, has now been observed in animals. The presence of these two major divisions of papillomaviruses in both human and monkey hosts strongly suggests that this diversification predated the evolutionary split between monkeys and apes. This would imply that at least two different groups of papillomavirus have evolved separately in their respective primate hosts for more than 22 million years with only moderate sequence changes since their genesis. The closest relatives of human papillomaviruses are the papillomaviruses that infect chimpanzees, our closest living relatives. And the next closest relatives of human papillomaviruses infect our next closest relatives, the gorillas. It is therefore likely that the common ancestor of humans, chimpanzees, and gorillas-an ape that lived some eight million years ago-carried its own strain of papillomavirus. The genealogy of HPV reflects the genealogy of our species. The oldest lineage of the virus is most common in living Africans, for example. Native Americans descend from East Asians, and their viruses share the same kinship.
Herpes: The Herpes virus is an example of how it has evolved along with humans. Approximately two-thirds of the human population is infected with at least one herpes simplex virus. The viruses are most commonly presented as cold sores on the mouth or lips or blisters on the genitals. Humans are the only primates we know of that have two herpes simplex viruses and researchers wanted to know why. They compared the HSV-1 and HSV-2 gene sequences to the family tree of simplex viruses from eight monkey and ape host species. It became clear that HSV-1 has been present in humans far longer than HSV-2, prompting the researchers to further investigate the origins of HSV-2 in humans. The viral family tree showed that HSV-2 was far more genetically similar to the herpes virus found in chimpanzees. This level of divergence indicated that humans must have acquired HSV-2 from an ancestor of modern chimpanzees about 1.6 million years ago, prior to the rise of modern humans roughly 200,000 years ago. In addition, some genes of this virus were found in bacteriophages, suggesting a common origin. The most common ancestor of all known Herpes viruses seems to have existed around 400 million years ago.
Hepatitis: Approximately one in every 12 individuals, or 500 million people worldwide, is living with chronic viral hepatitis. The Hepatitis B virus (HBV) specifically infects the liver cells of many primates (including humans), causing severe flu-like symptoms. Although most people fully recover, roughly 5 percent remain infected throughout their lives; acting as carriers who can infect others whilst also suffering a variety of serious liver diseases, including cancer. In fact, HBV is second only to tobacco amongst known human carcinogens. According to genomic analysis, scientists came up with 2 scenarios: the first ancestral HBV became trapped in avian DNA quite early: the common ancestor of all the bird species carrying this particular viral fossil lived about 82 million years ago. It infected birds way longer than it infected mammals by switching hosts. The other scenario was that the virus infected the first amniotes (ancestors of reptiles, birds, and mammals) and split into separate lineages more than 324 million years ago when the early bird and mammal lineages split.
Spanish flu: The 1918–1919 influenza pandemic killed more people than any other outbreak of disease in human history. One of the first casualties was the British prime minister and war leader, David Lloyd George. On 11 September 1918, Lloyd George, riding high on news of recent Allied successes, arrived in Manchester to be presented with the keys to the city. Female munitions workers and soldiers home on furlough cheered his passage from Piccadilly train station to Albert Square. But later that evening, he developed a sore throat and fever and collapsed. He spent the next 10 days confined to a sickbed in Manchester town hall, too ill to move and with a respirator to aid his breathing. Lloyd George, then aged 55, survived, but others were not so lucky. Recent scholarship estimates the death toll from 50 to 100 million dead. When it was reported in May 1918 that King Alfonso XIII was ill in Madrid, most people dismissed the Spanish flu as a joke. The main advice was to gargle with salt water and to isolate yourself until the fever had passed. However, these rules did not apply to munitions workers who were urged to “carry on” for the sake of the war effort.
When researchers analyzed the genome of the Spanish flu recently, they found that most of its genes were derived from a bird flu virus. The 1918 virus was also found to be the common ancestor of human and classical swine H1N1 influenza viruses, which continue to haunt us to the present day. Haskell County, Kansas was pointed as one of the places of origin of the Spanish flu, being close to the first outbreak at Camp Funston, Fort Riley. There the smell of manure meant civilization. People raised grains, poultry, cattle, and hogs. A rival theory, favored by the British virologist John Oxford, is that the pandemic began at Étaples, a huge British military camp an hour south-west of Boulogne. With accommodation for up to 100,000 soldiers, Étaples lay on a migratory bird flyway close to the Somme estuary and had all the necessary conditions for a spillover event: wild waterfowl, plus chickens and pigs, living in close proximity to men packed into airless barracks. Étaples also had several hospitals where soldiers whose lungs had been compromised by mutagenic gases deployed on the battlefield.
This flu had long term impact. Economist Douglas Almond has estimated that people exposed in the womb to the Spanish flu received less education, earned lower incomes, and were likelier to have disabilities than people who missed the pandemic in the womb. Its mortality rate ranged from 2% in developed countries to 6% in India where 18.5 million people perished.
Part 3: A peek into the viral universe
Let us trace the history of virology. Several hundred years ago in China, people inhaled powder from scabs of smallpox sufferers in order to immunize others. By the 1700s this was a well-known practice in the Arab world. In 1717 Lady Mary Wortley Montagu observed the practice in Istanbul and attempted to popularize it in Britain, by inoculating the Royal family. But her efforts never became popular because the methods still had a 2-3% chance of transmitting the disease. In 1721, Cotton Mather and Zabdiel Boylston wanted to inoculate citizens of Colonial Massachusetts. They started a program and continued to inoculate many volunteers, despite many adversaries in both the public and the medical community in Boston. As the disease spread, so did the controversy around Mather and Boylston. To make their point, Mather and Boylston used a statistical approach to compare the mortality rate of natural smallpox infection with that contracted by variolation. During the great epidemic of 1721, approximately half of Boston’s 12,000 citizens contracted smallpox. The fatality rate for the naturally contracted disease was 14%, whereas Boylston and Mather reported a mortality rate of only 2% among variolated individuals. This statistical analysis may have been the first time that comparative analysis was used to evaluate a medical procedure. While Cotton Mather is remembered as one of the most influential Puritan ministers and scholars of his day, he is also infamous in his role in the Salem witch trials. Scholars suggest that Mather’s dramatic descriptions of the devil’s activity upon children may have led to the first cry of witchcraft in Salem Village.
In 1796, Edward Jenner developed the much safer technique of vaccination using cowpox instead of smallpox. In 1876, Adolf Mayer described a condition of tobacco plants, which he called “mosaic disease”. He excluded the possibility of a fungal infection and could not detect any bacterium and speculated that a “soluble, enzyme-like infectious principle” was involved. In 1884, the French microbiologist Charles Chamberland invented a filter – known today as the Chamberland filter – that had pores smaller than bacteria and could filter them. In 1885, Louis Pasteur found a vaccine for Rabies and speculated about a pathogen too small to be detected using a microscope. In 1892, the Russian biologist Dmitry Ivanovsky used a Chamberland filter to study tobacco mosaic disease. His experiments showed that crushed leaf extracts from infected tobacco plants remain infectious after filtration. Ivanovsky suggested the infection might be caused by a toxin produced by bacteria, but did not pursue the idea. In 1898, the Dutch microbiologist Martinus Beijerinck repeated the experiments by Adolf Meyer and became convinced that filtrate contained new forms of infectious agents. He observed that the agent multiplied only in cells that were dividing and called it a “virus”.
The cause of Foot and Mouth disease in cattle was first shown to be viral in 1897 by Friedrich Loeffler. He passed the blood of an infected animal through a Chamberland filter and found the collected fluid could still cause the disease in healthy animals. In 1903 it was suggested for the first time that transduction by viruses might cause cancer. In 1908 Bang and Ellerman showed that a filterable virus could transmit chicken leukemia, data largely ignored until the 1930s when leukemia became regarded as cancerous. In 1911 Peyton Rous reported the transmission of chicken sarcoma, a solid tumor, with a virus, and thus Rous became “father of tumor virology”. The existence of viruses that infect bacteria (bacteriophages) was first recognized by Frederick Twort in 1911, and, independently, by Félix d’Herelle in 1917. As bacteria could be grown easily in culture, this led to an explosion of virology research. By 1928 enough was known about viruses to enable the publication of Filterable Viruses, a collection of essays covering all known viruses edited by Thomas Milton Rivers. We have come a long way since then. Let us now delve into what makes viruses special.
Mindboggling insights into the viral world
Sheer numbers: There are estimated to be 2*10 power 31 viruses in earth. That means 2 followed by 31 zeroes. Science writer Carl Zimmer tries to visualize this number, “As in over 10 million times more viruses than there are stars in the universe.” I calculated that if you were to stack one virus on top of another, you’d create a tower that would stretch beyond solar system, milky way galaxy and far beyond, to reach a height of 260 billion light years. This is enough to go to the edge of the known universe and come back 19 times. This number by the way is more than the number of sand grains in earth (8*10 power 18) or the number of drops in the world’s ocean (4.5*10 power 25). This number means that this is the most successful species ever, dwarfing even bacteria (5*10 power 30).
Diversity: Based on research that each species could harbor upto 58 different virus strains, the number of virus species could be at least 100,939,140. This includes the 1,740,330 known species of vertebrates, invertebrates, plants, lichens, mushrooms, and brown algae. To add context just 62,305 vertebrate species are known to exist.
Mobility: Researchers looked at a boundary layer in the atmosphere – the free troposphere. At this height, approximately 8,200 to 9,840 feet above sea level, viruses hitch rides on air currents and on particles of soil or vapor from sea spray, and travel thousands of miles from their point of origin. Researchers noted that “Every day, more than 800 million viruses are deposited per square meter above the planetary boundary layer”.
The human-virus connection: Not many will know that while there are 37 trillion cells in a human, a human body also has 38 trillion bacteria and upto 380 trillion viruses inside it. There is a growing body of research which shows the the human body is not just about human cells, but it should also factor in the bacteria and viruses which outnumber human cells by a 111:1 ratio. What are all these additional microbes and viruses doing?
The greatest massacres of all time: Bacteria and viruses have been involved in an evolutionary tug of war since the beginning of life. Viruses kill 40% to 50% of all bacteria in the oceans each day, which creates considerable organic material that floats to the bottom of the ocean (one billion tons of carbon each day). The carbon footprint of Humans is around 35 billion tons of carbon. This accomplishes in a month what all humans typically create in a year.
Bacteriophages are one group of viruses, found wherever bacteria exist. It’s estimated there are more than 1031 bacteriophages (aka phages) on the planet. That’s ten million trillion trillion, more than every other organism on Earth, including bacteria, combined. Each is evolved to infect a specific bacterial host in order to replicate — without affecting other cells in an organism. They cause a trillion trillion successful infections per second and destroy up to 40 percent of all bacterial cells in the ocean every day.
Research has shown that if a single phage virus is added to a culture of 10 billion E.Coli Bacteria, within two hours, there are 10 trillion viruses and only 10 million E. Coli remaining. This means that 99.9% of the E.Coli have been killed. Bacteria so dread these phages that they decrease their virulence to the host cells in response to phage attacks. Research has shown that in human cholera outbreaks, bacteria which infected humans, mutated their DNA to decrease their ability to make humans sick, in return for increased defense against phages. Hence phage treatment using the ability of viruses to scare bacteria into a meeker version of themself, may be a valuable alternative to antibiotics in the fight against harmful bacteria, in the future.
Reproductive ability: As science columnist Carl Zimmer writes “If you get sick with the flu, for example, every infected cell in your airway produces about 10,000 new viruses.” Within hours viruses can make millions or even billions of copies of itself. “The total number of flu viruses in your body can rise to 100 trillion within a few days. That’s over 10,000 times more viruses than people on Earth.” They also take advantage of the mammalian sneeze reflex to multiply. When an infected person sneezes or coughs more than half a million virus particles can be spread to those close by.
Part 4: The deepest questions of life
Is there a definition of life? Right from childhood, we are brought up to think that it is easy to differentiate living from non-living. Living beings can see, hear, smell, taste and feel. Humans are alive, but a rock or a table is not. Easy human-centric answers are simplistic and don’t tally with facts. One definition defines life as “absorbs compounds from its environment,” “excretes waste,” and “grows”. While fire consumes “food,” excretes “waste,” metabolizes, moves, and grows, it is not life. This shows how fallible definitions are. For a long time, it was not clear if plants were conscious, because they did not have brains and memory. We know better now. Researcher Paco Calvo argues that “Plants can, not only learn and memorize but also make decisions and solve complex problems. Plants can anticipate competition for resources, growing differentially depending upon the future acquisition of minerals and water (Novoplansky, 2015). They exhibit self-recognition and territoriality (Schenk et al., 1999), being able to tell apart their own from others, directing their movements towards their targets of interest (Gruntman & Novoplansky, 2004). Plants can communicate aerially with members of their own kind and with members of other species. They can even communicate bioacoustically, making and perceiving ‘clicking’ noises (Gagliano et al., 2012). Some plants can tell vibrations caused by predators apart from innocuous ones (wind or the chirps of insects), eliciting chemical defenses selectively (Appel and Cocroft, 2014). In a sense, plants can see, smell, hear, and feel (Chamovitz, 2012).”
Philosophers introduced abstract concepts like soul and inner spark, which is ultimately a fantastic and imaginative hypothesis. Researchers have identified 123 definitions of life, and condensed the finding as “Life is self-reproduction with variations.” NASA too came up with its own definition “”A self-sustaining chemical system capable of Darwinian evolution.” NASA did accept that its definition wouldn’t apply to viruses. Viruses typically do not have DNA. They are dead the entire time until they meet a host cell. They cannot reproduce by themselves. Even now, many scientists who look at the 3 kingdom framework do not accept viruses as living. But recently discoveries are turning these theories upside down.
Around 2003, a giant virus – Mimivirus was discovered on a French cooling tower which contained 979 genes—as many as some bacteria. Remember humans have 2000-25,000 genes. Megavirus was discovered in 2010, off the coast of Chile. It had 1120 proteins. In 2013, the Pithovirus was discovered. It possesses some replication machinery of its own. Another virus discovered in 2013, Pandoravirus has a staggering 2500 genes, seemingly heralding a new class of viral life. “More than 90 percent of its genes did not resemble anything else found on Earth,” noted a researcher.
Prions are harmless proteins that are present in mammals and birds. But these proteins are in an abnormal form, and once they enter the human brain, they are capable of causing severe brain infections. As soon as prion finds their way into the brain, they cause normal proteins to turn into abnormal ones. Then afterwards, they soon multiply causing severe infection in the brain. Due to this infection, some holes appear inside the brain that can only be treated by incineration. Some of the diseases caused by prion are Mad Cow Disease, Scrapie in sheep and goat, chronic wasting disease in deer and elk, Kuru and Creutz-Jakob disease. If prions are living, then are proteins living too?
Professor Nigel Brown argues that” If a virus is alive, should we not also consider a DNA molecule to be alive? Plasmids–? What about prions?” BBC reporter Josh Gabbatiss argues “Maybe the things we think are essential are really just peculiar to life on Earth. After all, everything from bacteria to lions is derived from a single common ancestor, meaning that on our chart of life in the Universe, we only really have one data point.” And because the definition of life is extremely restricted by our personal experiences and ancestry, it will be very difficult if not impossible, to look for other living forms that could exist. How would Silicon-based life look like if Carbon was not around? We don’t know the answers. And it is possible that our search for extraterrestrial life could be doomed to failure because humans cannot fundamentally reimagine the constraints within which they exist. At the same time, there are contrarian scientists like Carol Cleland who feel that language is vague, and all terms face borderline cases. The fact that there are borderline cases – that we can’t come up with a precise cut-off – doesn’t mean there isn’t a difference. Hence, entities like viruses do not provide interesting challenges to definitions of “life.”
The origin of cellular life: Based on some clues, viruses may have influenced and even created life as we know it. The first clue is that the amount of diversity in viruses far exceeds the diversity found in cell based life. Like the fact that the huge human genomic diversity in Africa is strong evidence of the African origin of Homo Sapiens, viruses may be the fountain of life. In the words of virologist, Valerian Dolja “Where diversity lies, origin lies.” Secondly, viruses are also more diverse in reproduction compared to cellular life. A final argument is that some of the viruses that infect the three oldest domains of life – Archaea, Bacteria and Eukaryote, all share several of the same proteins, suggesting that they may have evolved the earliest life form emerged.
As posited by Patrick Forterre, one of the most famous virologists of our time, “If viruses were already present in the biosphere when the last universal common ancestor (LUCA) was living, one would expect to find some common features between viruses that now infect members of different domains. This is precisely the case. In particular, some archaeoviruses, bacterioviruses and eukaryoviruses share homologues in proteins and ATPases, suggesting that they all evolved from a common virus that existed at the time of LUCA of even before.” Other biologists go so far to argue that “the interaction of hosts with retroviruses, retrotransposons, and retroelements is one of the eternal conflicts that drive the evolution of life.”
It is hypothesized that viruses might have contributed to the formation of the first cellular life form, or that even the eukaryotic cell nucleus originates from an infection by a coated virus. In a research paper published in 2014 by the American society of microbiology, it was argued that “all life was initially a floating stew of genetic information, amino acids, and lipids. The earliest pieces of genetic material were likely short pieces of RNA with relatively few genes that often parasitized other floating bits of genetic material to make copies of themselves. Over time, the parasitic genetic elements remained unable to replicate on their own and evolved into modern-day viruses that mooch off their cellular hosts. The genes they parasitized began to evolve different types of genetic information and other barriers to protect themselves from the genetic freeloaders, which ultimately evolved into cells.” In short, the explanation towards the formation of cellular life (including the universal common ancestor of all kingdoms of life) could probably have begun with the parasitic behavior of virus particles. This interestingly could make selfishness the original sin or virtue, as one would look at it. But there is also a different perspective here. Virologist, Patrick Forterre has suggested that viruses don’t steal genes from cells but rather that cells steal genes from viruses. Viral genomes are full of genes encoding proteins whose function probably is to regulate the interactions between viruses and their hosts. They have no similarities or homologues in cellular genomes. Unlocking the secrets of viral genomes could well unveil functions of genes never seen before and the secrets of life we never knew earlier.
Part 5: Coming to terms with the virus within
The standard model of a virus is that it is non-living, is a strand of RNA and is inherently destructive. While some parts of these descriptors may be correct in some scenarios, it would not be wise to tarnish all viruses with the same brush. Because our understanding of viruses is pretty recent, we have yet to wrap our mind around it and misconceptions remain. Recent scientific discoveries are challenging our basic assumptions.
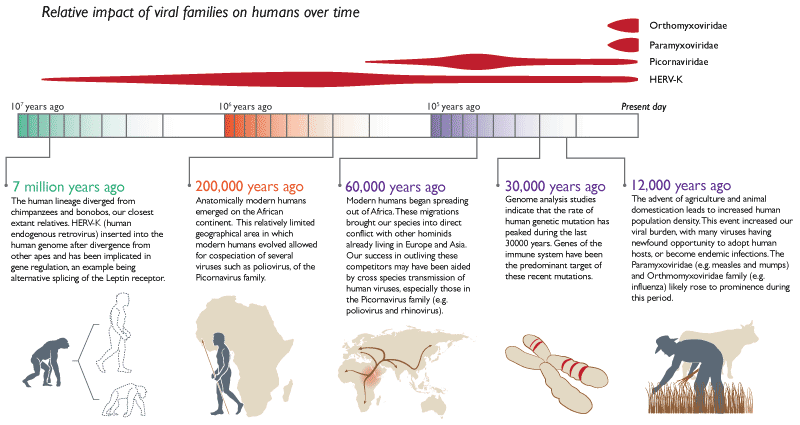
Connecting humans and viruses: We share around 9% of our DNA with viruses. Called human endogenous retroviruses (HERV), these trace their origin from retroviruses. Unlike viruses, retroviruses, however, use a slower, stealthier approach. After entering the cell, the retrovirus uses an enzyme called reverse transcriptase to turn its RNA into DNA before making its way to the nucleus. Once in the nucleus, it inserts its DNA into the host’s genome. The virus, however, wasn’t actively transmitted from animal to the other. Instead, a retrovirus had embedded itself in the germline and passed from parent to child. While such insertions typically happened millions of years ago, in the year 2000 researchers saw this happening with Koalas right in front of their eyes. This is remarkable considering that retroviruses have introduced themselves only 30-40 times into a typical animal genome in the past 60 million years.
Oxford Professor Aris Katzourakis, remarks “It (HERV) is changing how we think of ourselves as a species. Such an intimate interaction between ourselves and these viruses, and exchanging DNA that’s useful for us, has really molded how we’re now thinking of ourselves as a dynamic soup of DNA that’s now infiltrated by viruses,”
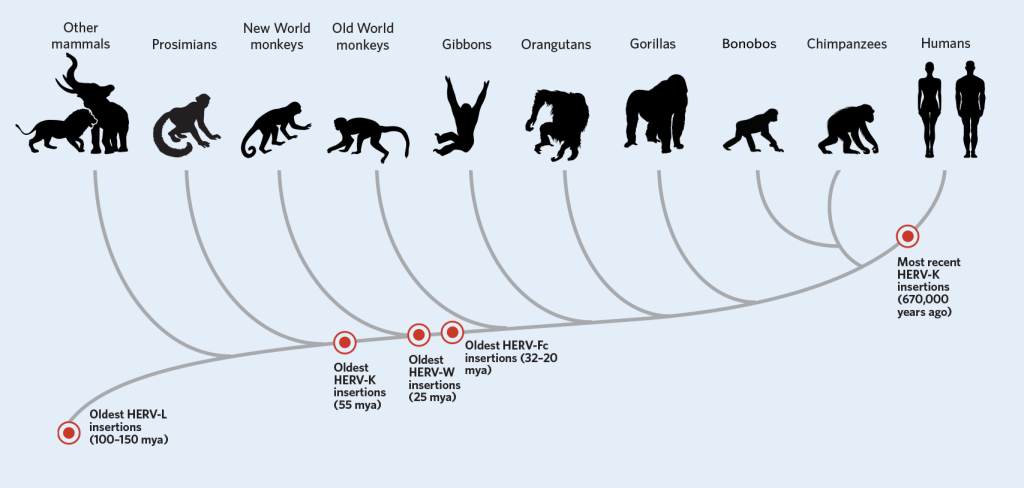
Let us begin by discussing HERV-K. According to research, HERV-K may also have played an important role in separating some of the first humans from their primate ancestors by making small adjustments in when certain genes were switched on or off. It activates key genes that help transform a single cell into a fully-formed infant. These HERV-K viral particles and proteins also help protect the tiny ball of cells from being infected by other viruses. HERV-H plays an instrumental in switching embryo cells into adult cells and vice-versa. A protein Syncytin is produced only by certain cells in the placenta, and it directs the formation of the cellular boundary between the placenta and maternal tissue. When scientists looked closer at the DNA sequence of syncytin, they found that it was nearly identical to a viral protein called env that caused the virus to fuse with its host cell. In the placenta, syncytin performed helped the fetus fuse with its mother. Humans aren’t the only species with a placenta, however. All mammals have placentas, including marsupials and egg-laying mammals. Although all of these mammals have a syncytin gene, they don’t all have the same syncytin gene. The syncytin produced by mice is completely different from the two syncytins found in humans and other primates. At numerous points in mammalian evolution, symbiotic retroviruses entered the genome and steered different groups of mammals along different evolutionary paths.
Transposons: 44% of all genetic material in humans is made of transposable elements also called transposons or jumping genes. These molecular parasites behave very much like viruses and ensure their own replication at every cost to the host cell. Just one transposon, the ALU sequence alone is estimated to make up 15–17% of the human genome. ALU insertions have been implicated in various types of cancers, sarcoma, hemophilia, diabetes and Alzheimer’s disease. At the same time, ALU insertions have also helped with the evolution of color vision in primates. Similar double agent roles of transposons include helping animal cells get immunity from microbes and also helping microbes get immunity from antibiotics. Paradoxically, transposons have played a key role in the formation of the placenta, formation of embryos, DNA methylation and aging.
Polintons: Also called Mavericks, they are DNA transposons that are widespread in cellular genomes. Polintons were the first group of cellular double-stranded DNA viruses to evolve from phages and that they gave rise to most large DNA viruses in cells like the Adenovirus and various other selfish genetic elements. Plasmids are very important component of all cells in the animal kingdom including animals and humans. Plasmids come from Polintons, the adenovirus ancestors. An old Polinton somehow left the nucleus with the new proteins and then evolved into Magavirales and these plasmids in the cytoplasm.
It is estimated that Adenoviruses which emerged from Polintons had started to coevolve with the vertebrates 450 million years ago, before the divergence of fish from other vertebrates. The fact that they exist in the genomes of protists, fungi, and all animals shows that Polintons are extremely ancient, with research indicating at least a billion years old. Evidence of even older ancestry comes from the fact that Polintons make proteins that are similar to the Major and Minor Capsid Proteins. These proteins construct the complex icosahedral geometric forms of the common double-stranded DNA virus covers for all three cellular kingdoms. The Polinton major capsid protein is similar to the large Megavirales family of viruses. This connects Polintons with the largest viruses and with smaller double-stranded viruses.
Let me close with a funny and thought provoking discussion. Humans are not only part virus but also part banana. Researchers from Cambridge reveal that while we share around 50% of our DNA with our parents, we share 50% of our genes with bananas. This interesting conundrum is resolved when you understand that genes – the regions of DNA that code for proteins – only make up about 2% of your DNA. “So sharing 50% of our genes with bananas means we only actually share 1% of our DNA with them. But animals and plants share a common ancestor – a single-celled life form which probably lived about 1.6 billion years ago. The genes that we share with bananas would have been present in that ancestor, and have been passed down to all animals and plants alive today. And the reason that we’ve kept these genes, is that they’re involved in fundamental cell processes – like making energy and repairing damage. Just like that single-celled ancestor, and our banana relatives, we need these processes to survive – and so we share half of our genes, but not half of our DNA with bananas.”
“The boundaries between organisms are a bit more merged now, a bit more shadowy. We need to break down those boundaries,” says University of Queensland virologist Paul Young. “The more we look, the more we find overlap.”
Notes:
- This article is heavily sourced and is meant to be thought-provoking and derivative.
- Size of a bacteriophage is =8000 nm = 8*10^-6 m. Assume there are 210^31 viruses, viruses occupy 2.5^10^24 sq m= 2.6*10^11 light-years = 260 billion light-years. Given that the universe is 13.7 billion light-years old, viruses can travel from the earth to the edge of the universe and back 19 times.
- Cover picture credit: Ultrastructural morphology of Coronaviruses, created at the CDC https://phil.cdc.gov/Details.aspx?pid=23312
Heavily researched. Some questions to ponder – where the answer to cure will come from? Will it be bats, pangolins, or humans. While we have lots of research on bats, the pangolin link has not been fully explored. Thanks for the virulent article.
LikeLike